
In the fall of 1950, a Halloween nightmare unfolded. Something in candy was making kids sick with severe diarrhea and abdominal cramps.
The event was a wake-up call for the Food and Drug Administration, launching it on the path of more oversight of food chemicals.
Today, 75 years later, food chemicals and dyes are back in the spotlight. Some members of Congress may soon introduce a bill that could repeal state laws that protect people from toxic food chemicals. The legislation would also likely block states from enacting new food safety laws.
That’s a big problem because states are out ahead in efforts to make our food safer, not the FDA – even though that’s one of the agency’s key purported functions.
‘Definitely toxic’
Poisoned Halloween candy is a persistent urban legend. The actual culprit in 1950 was Orange Dye No. 1, a food dye derived from coal processing byproducts and approved for use in 1907, along with six other dyes. To get a deep Halloween orange color, the tainted candy was made with large amounts of pure Orange No. 1 – in some cases, as much as 1,500 parts per million. The dye was making kids sick because it was toxic.
In response to the public outcry over Orange 1, the FDA tested that dye and two others, Orange Dye No. 2 and Red Dye No. 32.
The FDA’s findings for all three dyes were alarming – they were not “harmless substances,” as had been assumed, “but on the contrary are definitely toxic.” Orange 1 tests revealed “definite damage to various vital organs of the test animals, significant changes in body weight, and premature death.” Volunteers experienced diarrhea after eating one to eight pieces of candy.
All three dyes were banned in 1955.
Strengthening the FDA’s food oversight
Congress also took note. To investigate the use of chemicals in food and cosmetics in 1950, it formed a special committee, led by Rep. James Delaney (D-N.Y.).
The 1938 Food, Drug, and Cosmetic Act had given the FDA strong authority to protect the food supply from pathogens. But the agency wielded little power over the growing number of chemicals used as preservatives, thickeners, emulsifiers and sweeteners. Companies could use the new chemicals without FDA approval, and without assessing their long-term risks, which included harms such as cancer or reproductive and developmental harms.
The Delaney committee consulted experts and held public hearings. It found that the increased use of food chemicals “created a serious public health problem” and “no chemical should be permitted entry into the Nation’s food supply until its safety for use has been demonstrated beyond a reasonable doubt.”
The panel’s work culminated in the Food Additives Amendment of 1958 and the Color Additives Amendment of 1960. Both laws overhauled the FDA’s authority to keep us safe from food chemicals. They required a new chemical to get FDA approval before it could be used in food and put the burden on companies to prove chemicals were safe.
But today’s food chemical regulatory system is a far cry from what Congress envisioned in the 1950s and 1960s.
Real mass poisoning events like the Halloween scare of 1950 are rare. But the system fails to address more insidious threats, like increased risk of cancer, nervous and immune system harms, hormone imbalances, and even premature death.
The FDA has also been slow to act on food dyes. In 1990, it found that Red Dye No. 3, an ingredient in Halloween candy like candy corn and Laffy Taffy, can cause cancer. It banned several uses of Red 3, but none of its food uses, even though the FDA is required to prohibit food uses of carcinogens.
EWG and several other nonprofit organizations petitioned the FDA in 2022 to follow its own science and finally ban Red 3. But the FDA didn’t ban it until January 2025, nearly 35 years after first declaring it a carcinogen.
States take action
Roughly 75 years after some dyes caused mass illness, in 1950, some states are stepping up to address risks from dyes still on the market.
In 2024, California banned six artificial food dyes from school foods, after California’s Office of Environmental Health Hazard Assessment found strong links in some kids between exposure to the dyes and neurobehavioral issues.
Arizona, Louisiana, Utah and Virginia followed suit this year. West Virginia banned several artificial dyes from school foods, starting in August 2025, and from all foods starting in January 2028. Several major companies have announced they will phase these food dyes out of their products to comply with the West Virginia ban.
Broken system
Few food chemicals go through the FDA safety review and approval process created by the 1958 law.
Instead, most take advantage of a legal loophole that allows them to decide – sometimes without ever telling the FDA– that their chemical has been found to be generally recognized as safe, or GRAS.
An EWG analysis found that nearly 99% of new food chemicals get market access by exploiting the GRAS loophole.
Some companies decide to share their safety findings with the FDA as part of a voluntary process. But they’re allowed to withdraw a GRAS application at any time, demanding the agency stop its review of safety evidence. They then can proceed to use their chemical without further FDA scrutiny.
Examples of companies’ requests for the FDA to stop reviewing food chemicals’ safety
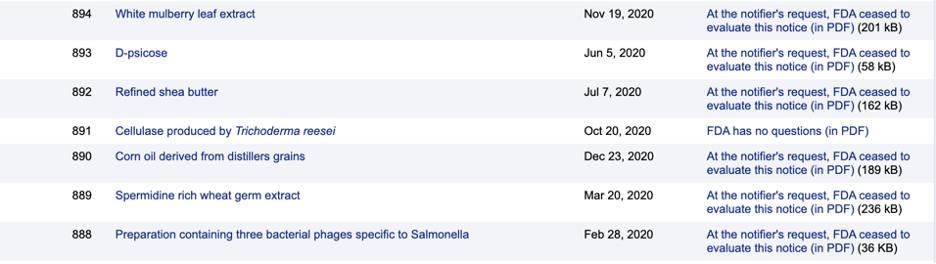
Source: FDA GRAS Notice Inventory
The GRAS loophole has allowed food chemicals of concern to enter the market. Examples include butylated hydroxyanisole, or BHA, and butylated hydroxytoluene, or BHT, which have been linked to reproductive harms and increased risk of cancer, and propyl paraben, which can alter gene expression and reduce fertility.
As a former FDA deputy commissioner acknowledged in 2014, “We simply do not have the information to vouch for the safety of many of these chemicals.”
The GRAS loophole is not the only way our food chemical safety laws are broken. Many food chemicals have been approved by the FDA, even though safety assessments have not been “meaningfully reviewed” in decades.
Titanium dioxide, historically used in Skittles and Starburst, was approved as a color additive in 1966 but has not been thoroughly analyzed for safety since. By contrast, Europe’s top food safety agency recently said it no longer considers the additive to be safe.
EWG and other nonprofit organizations petitioned the FDA to ban titanium dioxide in 2023, but the FDA hasn’t yet responded. Thankfully, the market isn’t waiting for the FDA. Walmart plans to prohibit use of titanium dioxide and dozens of other food chemicals in its store brand by 2027. Mars, the company that manufactures Skittles, announced in May that it phased out titanium dioxide from the candy at the end of last year.
Scientists also recently flagged potential immune system concerns about tert-butylhydroquinone, or TBHQ, a common preservative found in Pop-Tarts and other popular food. But the FDA has not meaningfully reviewed TBHQ for immunotoxicity since 1972.
Walmart also plans to ban the additive in its store brand, joining several other retailers including Albertson’s, Kroger, Whole Foods, Publix, and Hy-Vee.
In 2024, the FDA solicited public input about its reassessments of food chemicals. Earlier this year, the agency also released a rough outline for how it might prioritize chemicals for reassessment.
But these documents offer few details, leaving many questions about how, when, how quickly and under what kinds of guidelines the FDA will reassess chemicals of concern.
Protecting states’ rights
The absence of meaningful federal action highlights why it makes no sense to block states from protecting consumers even as the FDA drags its feet.
Until the FDA proves it can be trusted to police food chemicals, states should continue to drive change and make food healthier for consumers.



