
Thousands of everyday food products potentially could contain substances that carry unknown health risks, a new EWG analysis finds.
Although Congress intended for most food chemicals to be rigorously reviewed before being introduced into the market, the reality of food chemical review is far different. A flood of unregulated and potentially unsafe substances have been allowed in many products Americans eat.
Chemical and food manufacturers have rubber-stamped at least 111 food chemicals for use in numerous products, from cereal to snack bars, sports drinks and more.
Presence on the list of 111 food chemicals EWG identified does not mean a substance is harmful. But food companies have made their own safety determinations about these chemicals, without notifying the Food and Drug Administration and often while keeping details about their safety a secret, according to the analysis.
Of the 111 substances identified in the report, 49 chemicals are listed as ingredients in thousands of food products in the Branded Foods Database, a global compendium of food products and their ingredients maintained by the Agriculture Department, based on label data provided by commercial food brands.

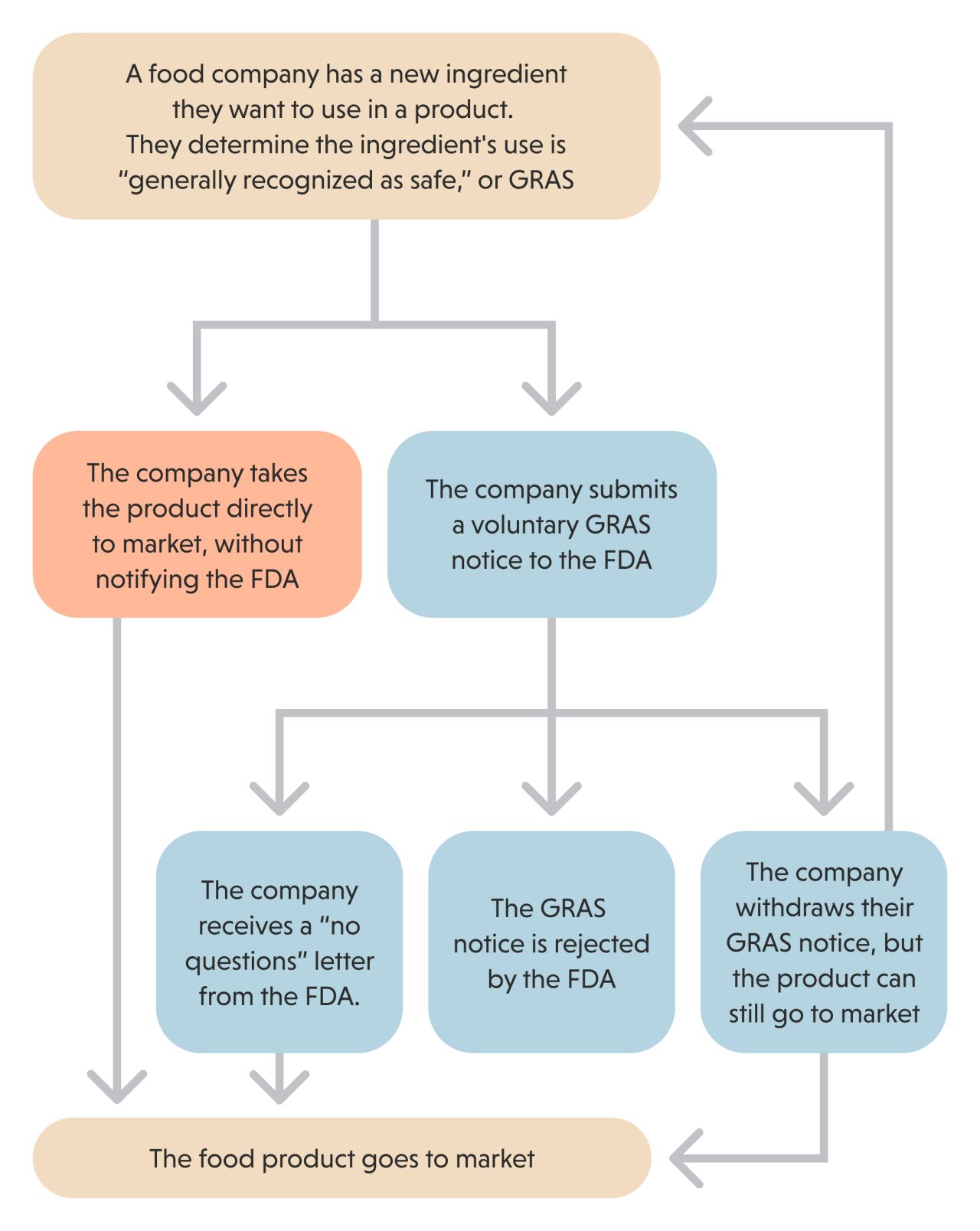
The analysis reveals how food and chemical companies have exploited a loophole in federal law in order to certify that their own new additives are “generally recognized as safe,” or GRAS. These chemicals are sometimes referred to as “secret GRAS,” because companies determined their safety without notifying the FDA. This is completely legal.
For consumers, this means a wide range of popular food and drink items may contain chemicals the government has never reviewed for safety. Any potential health risks of these secret GRAS chemicals may be known only to the companies that make them.
| Product name | Ingredient | Number of products | Types of food | Department of Agriculture link |
|---|---|---|---|---|
| Teavigo® | Green tea extract*, ** | 901 | Water, powdered drinks, smoked fish, candy, chewing gum, energy and granola bars, sausages, juice drink blends, yogurt, seasoning, ice cream | USDA Link |
| Qmatrix® | Aloe vera | 457 | Fruit and vegetable juice, nectars and fruit drinks, plant-based water, tea bags, soda, yogurt | USDA Link |
| Realturkeytail™ – turkey tail mushroom extract | Mushroom extracts | 428 | Coffee, energy, protein and muscle recovery drinks, powdered drinks, milk additives, seasoning, bottled tea, cheese, dips, snack, energy and granola bars, sauce, frozen dinners, entrees and sides | USDA Link |
| Roche lycopene | Lycopene** | 338 | Soda, fish and seafood,; tea bags, milk, chewing gum and mints,; cake, cookies and biscuits,; popcorn, peanuts, seeds and related snacks,; alcohol, sauces, water, salad dressing | USDA Link |
| Cognigrape™ | Anthocyanins and proanthocyanins | 219 | Seasoning mixes, marinades, tenderizers, candy and confectionary’ syrups and molasses, fruit and vegetable juice, nectars and fruit drinks, canned fruit, water, soda, dips and salsa, cake, cookies and cupcake mixes | USDA Link |
| Svetol® | Green coffee bean extract | 156 | Coffee, chocolate, soda, fruit and vegetable juice, nectars and fruit drinks, snack, energy and granola bars, powdered drinks, sport drinks | USDA Link |
| BioVin® 20 | Grape skin extract** | 151 | Soda, fruit and vegetable juice, nectars and fruit drinks, cooking sauces, candy, canned fruit, chocolate, dips and salsa, salad dressing | USDA Link |
| Chocamine® | Cocoa extract, theobromine** | 152 | Granulated, brown and powdered sugar, snack, energy and granola bars, coffee, powdered drinks, bottled tea, candy, cereal, ice cream, frozen yogurt, cookies and biscuits | USDA Link |
| Dimagnesium malate | Amino acid chelate | 113 | Powdered drinks, meal replacement supplements, specialty formula supplements, alcohol, energy, protein and muscle recovery drinks, milk additives, frozen desserts, ice cream and frozen yogurt, Mexican dinner mixes, snack, energy and granola bars | USDA Link |
| Cinsulin | Cinnamon extract ingredient | 104 | Honey, vegetable and cooking oils, tea bags, plant-based water, cereal, canned fruit, dessert toppings, popcorn, peanuts, seeds and related snacks, alcohol, milk, powdered drinks, fruit and vegetable juice, nectars and fruit drinks, pudding and custards | USDA Link |
| *The green tea extract composition is unknown; it could be a mixture or a single purified component like epigallocatechin gallate or EGCG. A search for “EGCG” produced 34 results; it was listed in foods such as soda, energy drinks, energy, protein and muscle recovery drinks. | ||||
| **Companies manufacturing some forms of these ingredients have notified the Food and Drug Administration, meaning they are not “secret,” but other companies have created other forms without notifying the FDA. There may been important differences in concentration, extraction method and other conditions of use among different forms. It is impossible to tell from the label whether a product uses an ingredient for which the FDA has received notice or whether it uses a version about which the company has made its determination without notifying the FDA. | ||||
Some of these “secret” chemical ingredients have been in the food supply for decades. For example, EWG found aloe vera, which a company declared safe for use in food in 2010, in more than 450 products.
The FDA banned the use of aloe vera in over-the-counter laxatives in 2002 over concerns about the substance’s links to cancer, kidney harm and low potassium blood levels. But the FDA has not acted to restrict its use in foods.
The European Union banned some kinds of aloe extracts and other botanical ingredients from food in 2021, but a court overturned the ban in 2024, finding that the European Food Safety Authority’s jurisdiction did not extend to “preparations” of ingredients such as extract methods. The court also said the EFSA failed to show how much certain component materials of concern would end up in food at unsafe levels.
Generally recognized as secret
A company’s declaration that its chemicals are generally recognized as safe allows it to dodge FDA pre-market review. Companies can add their new substances to prepared meals, snacks, candy, drinks and other food products without FDA restrictions or oversight. Sometimes the companies have reviewed the substances poorly or without transparency.
When Congress created the GRAS exemption in 1958, it intended the loophole to apply largely to basic, familiar ingredients such as salt, baking powder, and yeast. The idea was for the FDA to review and decide whether to approve most other chemicals before they entered the marketplace. But over time, companies began to exploit the loophole rather than submit their products to the FDA for review.
In 1997, the FDA made it even easier for companies to effectively police themselves by exploiting the loophole. It created a voluntary notification system, which lets companies decide whether to notify the FDA about new chemical ingredients they have determined are safe for use in food.
Under this system, even if a company voluntarily notifies the agency about its new chemical, it can change course at any time. If the FDA asks questions or raises safety concerns, for instance, the company can remove its submission from review and still sell it to manufacturers for use in food.
For example, gamma aminobutyric acid, or GABA, is a neurotransmitter that we all have in our bodies that, among other things, can help us fall asleep. Some companies manufacture GABA as a dietary supplement and sell it as a sleep aid and others have tried to add it to foods.
A Japanese company submitted a GRAS notice about GABA to the FDA in 2008, and again in 2015. The company withdrew both notices after the FDA raised concerns about:
- a lack of developmental and reproductive toxicity studies
- lack of information about how the body would process and eliminate GABA
- whether GABA could cross the blood-brain barrier
- estimated daily exposures potentially exceeding safe levels
- the use of unpublished studies
- possible impacts on the endocrine system
The company never submitted another notice addressing the FDA’s concerns. But GABA is still sold in supplement products such as these chocolates marketed to help with sleep, mental balance, relaxation and stress relief.
An EWG analysis of voluntary notifications to the agency since 2000 shows that nearly 99% of new food chemicals have exploited the GRAS loophole to come onto the market. Companies sent the agency 863 notifications for new substances between 2000 and 2024, though companies may have determined that an untold number of other chemicals were GRAS – without notice to the FDA.
In the same period, the FDA reviewed 10 new food additives and approved only seven through its pre-market review system.
The system is broken
This broken process has allowed hundreds, if not thousands, of chemicals of unknown safety to be added to food products – without FDA oversight or public awareness about the potential health risks.
For example, in 2022, nearly 400 people became seriously ill after consuming products made with tara flour, a secret GRAS ingredient. The makers of tara flour never filed a voluntary notice with the FDA. The FDA became aware of tara flour’s potential safety issues only after people started getting sick. This incident highlights how the GRAS loophole leaves federal regulators in the dark until harm has already occurred.
Withholding mandatory disclosure to the FDA has not only opened the door to new and unknown food chemicals. It has also allowed companies to unsafely use chemicals that are already on the market.
When a company determines that something is GRAS, that status is supposed to apply to certain uses of the substance only, often under specific conditions relating to concentration and temperature.
For example, caffeine is considered GRAS for use in colas. But in the early 2000s, non-cola beverage brands began to add caffeine to highly alcoholic beverages. The brands – including Four Loko – secretly determined that caffeine was also GRAS for use in these alcoholic drinks But alcohol and caffeine together make a combination that has allegedly led to serious injuries and even death.
Truly ‘generally recognized as safe’?
Manufacturers of the chemicals EWG identified claim they are GRAS, but some likely don’t legally qualify.
Under the FDA’s GRAS regulations, a chemical cannot be “generally recognized as safe” unless at least three things are publicly disclosed and scientifically recognized: 1) identity, 2) chemical composition, and 3) the basis for its safety determination.
A company GRAS determination should be based on “the same quantity and quality of scientific evidence” as a chemical going through the FDA’s more rigorous premarket approval process. That means companies are supposed to estimate how much people are likely to consume and describe what is known about the chemical’s potential hazards.
The regulations define safety as “a reasonable certainty … that the substance is not harmful under the conditions of its intended use.” This means if there’s doubt about the safety of the ingredient, it cannot be considered GRAS.
Safety determinations must consider the effects of exposure to the substance combined with any “chemically or pharmacologically related” substances in the diet. None of the companies marketing the chemicals EWG reviewed publicly disclosed any analysis of their total effects.
Safety must be “generally recognized.” That means there should be publicly available data. This data should usually be published in a peer-reviewed scientific journal, though studies may be published on a company’s website instead, according to the FDA.
The ingredients EWG reviewed rarely pointed to or provided published, peer-reviewed studies or any other public documentation establishing a basis for the safety of their “self-affirmed GRAS” ingredients.
Many GRAS substances are also newly developed ingredients. By its very name, GRAS should be based on a “general recognition” of safety among experts. But by definition, novel ingredients have not been used long enough for any “general recognition” or scientific consensus to form.
Most common uses of secret GRAS chemicals
EWG’s analysis found secret GRAS chemicals in a wide variety of foods. Three trends stand out: Many of the secret GRAS chemicals were either plant-based extracts, used as dietary supplements, or used as new ingredients to boost the protein content in foods.
Extracts
Whole foods like mushrooms, grapes and green tea are commonly associated with a diet rich in a diversity of nutrients and antioxidants. Because many consumers seek these properties, companies have identified substances responsible for these benefits and extracted them from their source.
But the extracted, purified components are much more concentrated than the unprocessed plant and could be harmful. Of the 49 ingredients EWG found in food, nearly half – 22 – were extracts. Although some are used to add flavors or antioxidants to food, many are simply marketed as ingredients, with little detail about their origin, composition or technical effects.
The analysis identified various forms of extracts among secret GRAS ingredients, derived from grapes, lemon balm, hemp, green coffee bean, cinnamon, cocoa and others.
Once taken from the plant, the extract is manipulated through processing and other chemical ingredients may be added. The resulting industrially produced compounds may not have the same health benefits as the original food. Instead, they’re concentrated and sometimes used in amounts much larger than the body can handle – which could, in some cases, lead to toxic effects.
For example, consumption of green tea is associated with several health benefits, such as lower cancer risk and LDL cholesterol and impacts on weight loss. Credit for these effects often goes to compounds commonly found in green tea, called catechins, known for their antioxidant properties. Epigallocatechin gallate, or EGCG, is the most used catechin compound, because it is the most abundant in green tea and may function as an antioxidant.
But concentrated EGCG and other concentrated catechins may well be an entirely different matter, with different and possibly harmful effects.
Studies have linked purified EGCG to heart and brain defects, fetal leukemia, suppression of estrogen, and liver, kidney and intestinal toxicity. EGCG is used in concentrations much higher than typically encountered in green tea. The FDA recognized these toxic effects as far back as 2008.
Nonetheless, EGCG is commonly used in sports and energy drinks, sodas and powdered drinks. And our analysis found other types of green tea extract continue to be added to the food supply without any notice to the FDA. Or, as in one instance, the FDA received two notices for catechins from green tea extract, in 2007 and 2009, both of which the company withdrew.
None of these products disclose information about the concentration of EGCG extracted from green tea or the amount used in the product. That makes it impossible for individuals who consume these products regularly to know how much they are exposed to and whether those levels are safe.
Another ingredient of unknown safety used in food is certain varieties of mushroom extract. This substance is found in 428 products, including coffee, powdered drinks, seasoning mixes, canned fish, stir fry sauces, pasta, canned soup and dairy-free cheese.
Companies may use a variety of mushroom extracts from various kinds of mushrooms, which are extracted through different methods and by different manufacturers. Some companies have notified the FDA about their extracts, but others have made their determinations in secret.
Because mushroom extracts are often listed just as a “mushroom extract” on a food label, it’s nearly impossible for consumers to know which kind of mushrooms it was extracted from, which part of a mushroom has been extracted, and whether it is safe to consume. Consumers also cannot know whether the mushroom extract in their product is one the FDA knows about or whether it had determined in secret that it was safe.
At least one company announced in 2022 that it had determined its mushroom extract was safe but did not send a notice to the FDA.
In 2024, the FDA moved to ban a different variety of mushroom extract also determined in secret to be safe in food – because of concerns about the “potential for serious harm and adverse effects on the central nervous system.” That mushroom is known to cause hallucinations.
Despite the ban in food, this ingredient remains available for sale in dietary supplements.
Grape extracts are especially common. Our analysis found that some form of grape extract or grape skin extract is used in more than 150 products on grocery store aisles. These include seasoning mixes, candy, fruit juice, syrup, canned fruit, sparkling water, soda and yogurt.
Green bean coffee extracts are also common. A green coffee bean extract was marketed for weight loss in 2008. Its manufacturer affirmed its GRAS status for use in food without notifying the FDA. Our analysis found green coffee bean extract in 156 different types of coffee, energy drinks, snack bars, powdered drinks, sports drinks and sodas.
A number of factors affect an extract. Many companies create extracts and in a variety of forms. They may be produced using different kinds of solvents, target different components of the whole food, or contain different final concentrations.
The FDA has received notice and safety information about some forms of extracts but not others. In the same way, an industry trade association may have reviewed safety information and informally provided some data to the FDA on some forms, but not others. But none of that information is available on the label, making it nearly impossible for the consumer to parse details about the extract and whether its safety was determined in secret.
These extracts sound benign, and many likely are. But it’s disturbing that there is often no public information on their processing or potential safety concerns. Of the 10 most frequent GRAS chemicals found in USDA’s food ingredient lists, six are extracts.
Supplements
Of the 111 secret GRAS chemicals found in EWG’s analysis, at least 18 are marketed for use in dietary supplements, including bovine colostrum, coenzyme Q10, amino acid chelates, trace minerals, garcinia cambogia extract and beta-glucan.
Federal law requires new supplement ingredients to submit to a formal process known as a new dietary ingredient notification. Companies must notify the FDA at least 75 days before selling these ingredients in products.
The GRAS loophole creates a back door for supplements to bypass this critical safety check. If a supplement ingredient is also used in food, a company can declare it GRAS for that use and skip the FDA notification process altogether.
This omission threatens consumers with the risk of unknowingly ingesting unreviewed substances that may not be safe in either food or supplements.
In 2015, Quincy Biosciences submitted to the FDA a voluntary GRAS notice that it had determined apoaequorin, a protein in jellyfish, safe for use in foods like protein shakes. The supplement, better known by its brand name Prevagen, is extensively advertised as a memory aid.
Quincy’s 2015 voluntary GRAS notice came after the FDA had twice rejected the company’s ingredient notifications, in 2008 and 2012. The FDA cited the company’s failure to substantiate safety claims. The agency noted that a single serving of Prevagen “would be the equivalent of 400-800 lbs of raw jellyfish consumed.”
The company’s own GRAS filing to the FDA included 2,200 reports of Prevagen’s health harms, including memory impairment, anxiety, hypertension, headaches, dizziness, insomnia, nausea and diarrhea. There were also 26 other harmful reactions, representing mainly nervous system and cardiovascular problems.
When the FDA asked about Quincy’s voluntary notice, the company withdrew its submission. In December 2024, a court found Quincy misled consumers about Prevagen’s ability to improve memory. Today Quincy claims Prevagen has “achieved self-affirmed GRAS status” and continues to sell it for use in protein shakes.
This isn’t an isolated case. While Quincy notified the FDA about its GRAS determination, other manufacturers do not. The use of secret GRAS to bypass the FDA’s supplement review process is longstanding. A 2014 review by the Natural Resources Defense Council found that “almost all of the [secret GRAS] chemicals NRDC reviewed were also ingredients in dietary supplements.”
More than a decade later, EWG’s findings show the practice has not changed. Companies still use the GRAS loophole to leapfrog mandatory FDA supplement safety reviews, exposing people to potentially harmful ingredients, often without even telling the FDA.
Alternative proteins
Protein is having a cultural moment. Food companies race to meet consumer demand for high-protein products, adding it to all kinds of foods, including pancakes, snack bars and even coffee. So it’s no surprise that some of the newer secret GRAS substances we found were alternative new proteins the FDA has never reviewed for safety.
For example, in 2024 one company announced it had achieved “self-affirmed Generally Recognized as Safe (GRAS) status for its air-based protein,” an alternative to plant and animal proteins, to be used in foods ranging “from noodles to baked goods and beverages to meat, dairy, and egg alternatives.”
Another company touted in 2025 that it had achieved “self-affirmed Generally Recognized as Safe (GRAS) status for its mycoprotein ingredient” to be used in “plant-based alternatives, sports nutrition, ‘healthy snacking’, meat imitates, dairy imitates, biscuits and sweet baking, extruded breakfast cereals, and breads and crackers.”
Some of these companies indicated they planned to notify the FDA about their GRAS determinations. But they had not at the time of releasing this analysis. Because these alternative proteins have not been reviewed by the FDA, it is unclear whether they pose a health risk.
Some proteins are known to cause allergic reactions, including life-threatening anaphylaxis, in some people. But the proprietary fermentation systems used to produce them are often black boxes, with no public disclosure of potential chemical byproducts, additives or contaminants.
Without FDA oversight, it is difficult for consumers to know what they’re really eating.
States in the lead
For decades, because of the FDA’s failure to close the GRAS loophole, ingredients have been secretly identified as safe and added to food without FDA review.
Now states are stepping in to bring long overdue transparency and protections to the public.
New York legislators last year year introduced the Food Chemical Disclosure Act, which would require companies to report to the state any chemical they have declared GRAS. This change would bring critical transparency to the process.
If enacted, it would make the use of secret GRAS ingredients illegal in New York, providing the public and regulators with much needed information about what’s being added to the food supply.
Pennsylvania lawmakers have introduced a similar GRAS transparency bill as part of a broader “Healthy Pennsylvania” bill package.
States have also banned some of the worst ingredients in food. In 2023, California enacted the California Food Safety Act, a landmark U.S. law banning four food chemicals already prohibited in Europe: Red Dye No. 3, brominated vegetable oil, propyl paraben and potassium bromate.
A year later, the state enacted the California School Food Safety Act, banning six food dyes from food served in public schools.
Momentum is growing, with other states following suit. In 2024, more than a dozen states introduced bills. In 2025, more than 30 states introduced bills, and new laws passed in Arizona, Louisiana, Texas, Utah, Virginia and West Virginia.
Members of Congress are also weighing in. Sen. Edward Markey (D-Mass.) and Reps. Rosa DeLauro (D-Conn.) and Jan Schakowsky (D-Ill.) have introduced legislation that would close the GRAS loophole and require the FDA to reassess chemicals of concern. Rep. Frank Pallone (D-N.J.), ranking member of the Energy and Commerce Committee, has also introduced legislation addressing the GRAS loophole.
As state protections grow, any potential federal legislation must continue to allow states the authority to continue to lead on food safety.
What the FDA should do
In March 2025, the Department of Health and Human Services acknowledged what scientists and advocates have warned for decades: The FDA’s system for overseeing food chemicals, particularly the GRAS loophole, is deeply flawed.
But rather than propose meaningful reform, the announcement merely pledged to “take steps to explore” revising a system that has been broken for more than 60 years. The FDA has indicated it may propose a rule soon, but few details are available. The rule is currently under review with the White House Office of Management and Budget.
To truly address the GRAS loophole, the FDA should set clear regulatory guidelines, with expert input. Any updated GRAS rule should ban secret determinations, prohibit the use of GRAS for novel substances, and block the use of GRAS for substances with evidence of health problems, including cancer, developmental, reproductive, metabolic, behavioral harms and chronic disease.
The FDA should stop letting companies withdraw a GRAS submission once the agency has raised safety concerns. And companies should not be allowed to market, sell or use that chemical as GRAS.
The scientists who make GRAS determinations on behalf of companies must be truly independent, with no conflicts of interest.
The FDA should also do much more to enforce existing GRAS regulations. It should revoke the GRAS designation of ingredients it deems unsafe and take them off the market. The agency can then notify manufacturers, importers, distributors and retailers that the substance is no longer GRAS.
The FDA should also enforce the requirement that companies base GRAS designations on publicly available data and vital health information that is scientifically recognized. This should include how a substance is used in food, its potential cancer and chronic disease risks, and its cumulative health effects as well as potential health effects from any biologically-related substances.
In addition to addressing the GRAS loophole, the FDA must also address the thousands of chemicals already in use. It must launch a scientifically rigorous post-market review program to reassess chemicals, including GRAS ingredients, that have remained on store shelves for decades without modern safety review. And Congress must fund FDA programs so it can complete this work.
In the absence of more concrete action from the FDA, states will – and should – continue to drive change for safer food additives and a more transparent system.



